Understanding TNF Inhibitors and Their Role in Autoimmune Care
If you live with an autoimmune condition, you’ve likely heard doctors mention "biologics." Among these powerful treatments, TNF inhibitors stand out as a primary defense against chronic inflammation. These medications target a specific protein in your body called tumor necrosis factor alpha. When left unchecked, this protein sends aggressive signals to your immune system, causing it to attack healthy tissues instead of fighting off viruses or bacteria. By interrupting this signal, these drugs can stop joint destruction, heal skin lesions, and reduce gut inflammation.
However, how they work isn't just about blocking one molecule. It involves complex interactions between your cells and signaling pathways. Understanding the difference between the available options, their mechanisms, and the necessary safety screenings can help you navigate treatment decisions with confidence. This guide breaks down exactly how these biologics function, which ones fit different conditions, and what you need to monitor while taking them.
The Biological Mechanism Behind TNF Inhibition
To grasp why these drugs work, you first need to understand the villain in the story. Your immune system uses chemical messengers known as cytokines to communicate. One of the most influential cytokines is TNF-alpha. Think of TNF-alpha as the commander-in-chief of inflammation. When your body detects injury, activated macrophages release TNF-alpha, which then binds to specific receptors on the surface of other cells, primarily TNFR1 and TNFR2.
This binding triggers a cascade of events. It activates signaling pathways like nuclear factor-kappa B, which leads to the production of more inflammatory chemicals such as interleukin-1 and interleukin-6. In a healthy response, this process is temporary. For someone with rheumatoid arthritis or Crohn's disease, however, this loop gets stuck in the 'on' position. The constant presence of TNF-alpha keeps immune cells marching through your joints or gut lining, leading to pain and tissue damage.
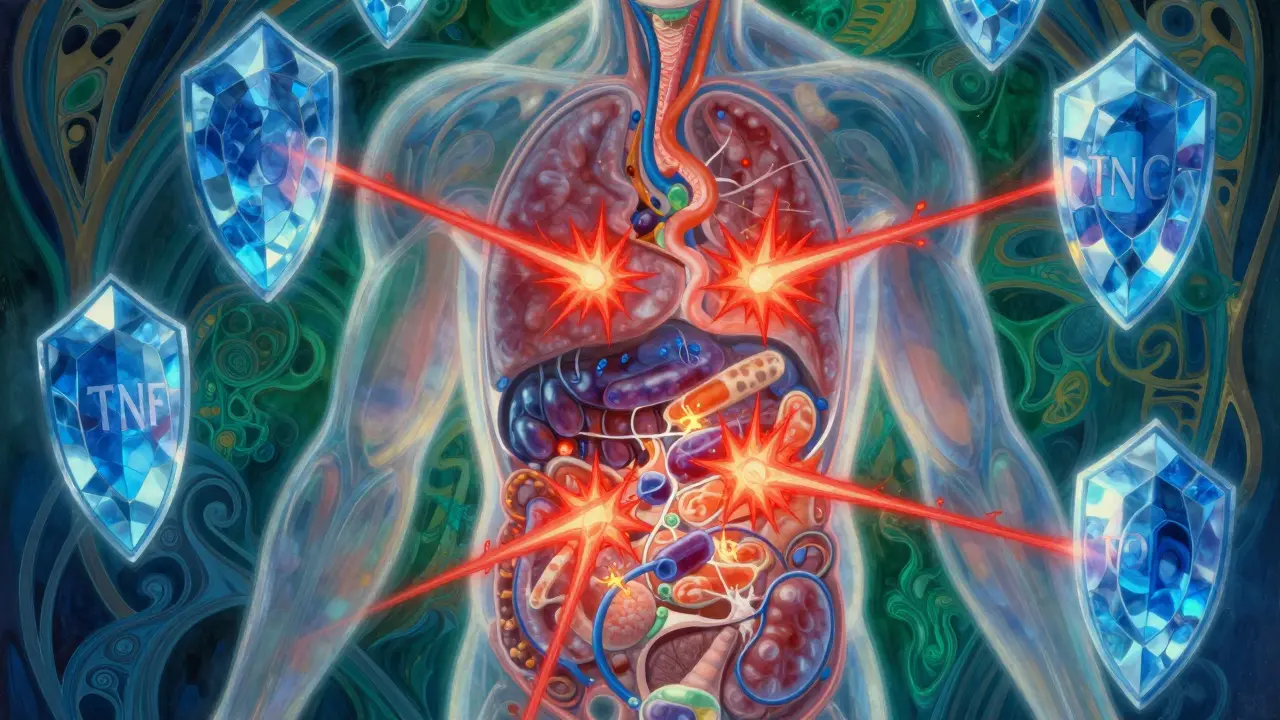
TNF Inhibitor Therapy targets this process by preventing TNF-alpha from binding to its receptors. Imagine placing a shield over the lock so the key cannot turn. There are two main ways these drugs achieve this blockage. Some act like decoys, floating around in the bloodstream to catch free-floating TNF before it reaches a cell. Others attach directly to the receptor sites on cells to prevent binding. Recent research published in the Journal of Leukocyte Biology highlights that beyond simple neutralization, these drugs also influence oxidative stress mediators circulating in the blood.
A critical nuance involves the blood-brain barrier. While these drugs are effective at stopping peripheral inflammation, they generally cannot cross the blood-brain barrier. This has implications for safety monitoring, particularly regarding neurological side effects. Studies in JAMA Neurology suggest that exposure to TNF inhibitors may be associated with increased risk for inflammatory CNS events because the drug cannot regulate TNF levels within the brain itself, potentially leading to localized imbalances.
FDA-Approved Agents and Structural Differences
Not all TNF inhibitors are built the same way. The United States Food and Drug Administration has approved five specific agents for autoimmune conditions, each with distinct structural features that influence how they interact with your biology.
| Drug Name | Structure Type | Target Specificity | Administration Frequency |
|---|---|---|---|
| Infliximab (Remicade) | Chimeric monoclonal antibody | Soluble and transmembrane TNF | IV infusion every 4-8 weeks |
| Adalimumab (Humira) | Fully human IgG1 antibody | Soluble and transmembrane TNF | Subcutaneous injection every 2 weeks |
| Etanercept (Enbrel) | Fusion protein (Receptor-Fc) | Primarily soluble TNF | Subcutaneous injection weekly |
| Golimumab (Simponi) | Fully human IgG1 antibody | Soluble and transmembrane TNF | Monthly injection or IV |
| Certolizumab pegol (Cimzia) | PEGylated Fab fragment | Soluble TNF only | Subcutaneous every 2-4 weeks |
These structural differences matter clinically. Monoclonal antibodies like adalimumab and infliximab contain the Fc portion of the antibody. This allows them to induce a process called complement-dependent cytotoxicity. Essentially, they can flag specific immune cells for destruction by your body’s natural cleanup crews. This mechanism contributes to their ability to modulate the immune system beyond simple blocking.
In contrast, etanercept acts differently because it is a fusion protein combining the extracellular domain of the TNF receptor with the Fc portion of human IgG1. It serves as a soluble receptor decoy. Because it lacks the full heavy chain structure of a complete antibody, it typically does not trigger the same level of cell-mediated toxicity, though this might also limit its induction of apoptosis in certain cell types compared to monoclonal antibodies.

Clinical Efficacy and Patient Outcomes
Since entering the market in the late 1990s and early 2000s, TNF inhibitors have substantially improved outcomes for autoimmune diseases. Before their introduction, conventional disease-modifying antirheumatic drugs often managed symptoms without preventing long-term joint erosion. Now, clinical trials consistently show that 50% to 60% of rheumatoid arthritis patients achieve significant symptom improvement with TNF blockers, compared to roughly 20% to 30% with older drugs alone.
However, success isn't universal. Approximately 30% to 40% of patients experience secondary failure, meaning the drug stops working effectively after an initial period of relief. The Cleveland Clinic reports that this often happens because the body develops anti-drug antibodies. These antibodies target the biologic medication itself, marking it for removal from the bloodstream before it can do its job.
Biosimilars have changed the economic landscape significantly. A biosimilar is a high-fidelity copy of an originator biologic once the original patent expires. For example, Amjevita was introduced as a biosimilar version of adalimumab. By 2022, these generics captured a significant portion of the market, increasing access for patients who previously found brand-name prices prohibitive. Despite concerns from some patients about switching to a biosimilar, major health organizations confirm they maintain similar efficacy profiles to the reference product.
Safety Protocols and Infection Risks
Because TNF-alpha plays a role in your body's defense against pathogens, suppressing it carries inherent risks. Your innate immunity relies on this protein to form granulomas-clusters of immune cells that wall off persistent infections. When you inhibit TNF, the barrier holding these infections dormant can weaken.
Before starting therapy, rigorous screening is mandatory. You must undergo a tuberculin skin test or interferon-gamma release assay to check for latent tuberculosis. Without prior suppression, reactivation risk jumps significantly. Patients on these medications have a 2 to 5 times higher risk of serious infections compared to the general population. The risk is highest shortly after initiation but persists throughout therapy.
Beyond tuberculosis, clinicians monitor closely for fungal infections and hepatitis B reactivation. Some users report injection site reactions, affecting approximately 20% to 30% of those using subcutaneous formulations. While usually mild redness or itching, severe reactions require medical attention. Furthermore, ongoing discussions in medical literature address paradoxical inflammatory events. Research suggests that inhibiting apoptosis of autoreactive T cells could theoretically lead to immune dysregulation in rare cases, sometimes manifesting as new autoimmune-like symptoms.

Practical Management and Daily Life
Living with a maintenance regimen requires logistical planning. Most TNF inhibitors involve self-administered injections, though infliximab requires clinic visits. The learning curve for managing needles typically takes one to two weeks with proper training. Many manufacturers offer support programs that include nurse access and copay assistance to mitigate costs.
Patient feedback varies widely. While some report life-changing improvements in mobility-going from barely walking to hiking regularly-others struggle with the psychological burden of frequent dosing. Missed doses can compromise effectiveness, making adherence crucial. It is vital to discuss vaccination strategies, specifically avoiding live vaccines while on immunosuppression, as the reduced immune surveillance makes such viruses dangerous.
Ultimately, TNF inhibitors represent a cornerstone of modern rheumatology and gastroenterology. They balance potent anti-inflammatory action with a defined safety profile. With continued research into selective inhibition of specific receptors, future versions may further refine this balance, sparing protective immune functions while eliminating harmful inflammation.
Frequently Asked Questions
Can TNF inhibitors cause liver problems?
Rarely, yes. Hepatitis B reactivation is a known risk, especially in carriers who were undiagnosed before starting therapy. Regular monitoring of liver enzymes is standard practice during treatment.
How long does it take for TNF inhibitors to start working?
Response times vary by individual and condition. Many patients notice changes within 2 to 4 weeks, but maximal benefit may take 3 to 6 months. Patience and consistent dosing are essential during this window.
Are there dietary restrictions when taking biologics?
There are no strict dietary bans, but maintaining a balanced diet supports overall health. Avoid unpasteurized foods since your immune response to foodborne bacteria is weaker. Hydration remains important for kidney health.
What is the difference between Etanercept and Adalimumab?
The main difference lies in structure. Etanercept is a fusion protein that targets soluble TNF, while Adalimumab is a monoclonal antibody targeting both soluble and membrane-bound TNF. Adalimumab may have additional cell-killing capabilities due to its structure.
Is it safe to get pregnant while on TNF inhibitors?
Many TNF inhibitors are considered relatively safe during pregnancy, but guidelines differ by specific drug. Always consult your rheumatologist before conception to weigh risks against the dangers of flares during pregnancy.






Comments
Calvin H
31 March 2026Great read though the formatting is terrible.
Michael Kinkoph
1 April 2026It is absolutely infuriating that this explanation glosses over the critical nuances of TNFR2 binding dynamics!! The general public deserves a deeper understanding of why chimeric antibodies differ structurally from fully human IgG1 variants!!! Ignoring these distinctions is negligent!!! Furthermore, the section on blood-brain barrier permeability lacks sufficient citation regarding longitudinal neurological risks!!! We must demand higher standards of informational integrity from such platforms!!!
Dan Stoof
2 April 2026I honestly believe this technology is a miracle for our generation!!! Imagine having a world where chronic pain no longer defines your daily existence!!! The progress we have made is nothing short of spectacular and vibrant!!! Every single patient deserves access to these life-altering solutions immediately!!! There is so much light waiting on the horizon for those who suffer now!!! Medication feels like a superpower when it stops the inflammation cascade effectively!!! My hope is boundless for everyone navigating this difficult journey forward!!! Keep fighting for yourself and never lose that inner spark!!! The future is bright for autoimmune warriors everywhere!!! We will conquer this illness together through unity and science!!! Remember to celebrate every small victory along the road to healing!!! Your body is resilient and capable of incredible recovery!!! Love and support are just as powerful as the medication itself!!! Always keep your head high and your spirits soaring high!!! The doctors are working tirelessly to protect us all!!! Stay magnificent and keep believing in better tomorrows!!!
Christopher Curcio
3 April 2026The mechanism involving nuclear factor-kappa B activation is crucial for understanding downstream IL-6 release. Patients often misunderstand the cytokine storm dynamic involved in rheumatoid synovitis progression. Soluble decoys versus transmembrane targeting dictates clinical response rates significantly. We must monitor anti-drug antibody formation carefully during maintenance dosing. Granuloma dissolution risks necessitate vigilant screening protocols for latent TB prior to initiation.
Brian Yap
4 April 2026Gday mates! Just wanted to say this info is pretty solid for what it is. Down under we get similar treatments here but prices vary wildly depending on where you live. Good to see people understanding the science behind their meds.
Ruth Wambui
5 April 2026It feels like pharmaceutical giants are pushing these expensive biologics to lock patients into eternal dependency cycles. They never mention the natural immune regulation methods that work without massive surveillance programs. Something is definitely being kept hidden about long-term liver enzyme toxicity spikes.
Vikash Ranjan
7 April 2026While I appreciate the data presented my personal experience suggests secondary failure happens faster than predicted. Many patients do not report adverse reactions until too late. I prefer traditional management strategies that do not compromise systemic defense mechanisms.
William Rhodes
8 April 2026Your attitude about the formatting is completely unhelpful to the conversation! We need to focus on the health benefits instead of complaining about layout design! Negativity kills progress and ignores the hard work researchers put into these drugs! Stop wasting time on petty critiques and support the community instead!
Angel Ahumada
9 April 2026people think they understand biology but they really do not grasp the fundamental philosophy of immune homeostasis it is deeper than receptors alone there is a spiritual connection to inflammation that modern medicine ignores completely while chasing patents the soul of the patient is neglected by these reductionist models we must look inward
RONALD FOWLER
10 April 2026Thanks for sharing this info. It helps to know the options available. We should all support each other through the process.